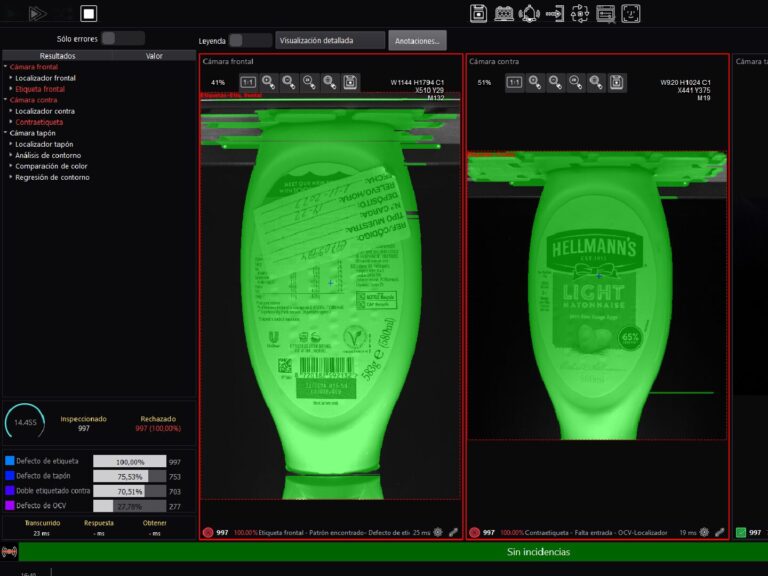
In the pharmaceutical industry, maintaining impeccable quality in packaging is not just a regulatory requirement. It is a fundamental necessity for patient safety and product efficacy. Any compromise in packaging integrity can lead to contamination, reduced effectiveness, or even severe health risks. This is why quality assurance in pharma manufacturing plays a critical role in every stage of production.
One of the key technologies ensuring this high level of quality is pharmaceutical rejection systems. These advanced systems are designed to eliminate defective products, ensuring that only safe, compliant, and high-quality medications reach consumers. By leveraging automation, vision inspection, and precision-based rejection mechanisms, pharmaceutical manufacturers can minimize human error and maximize efficiency.
A leading innovator in this field is E2M COUTH, a company specializing in state-of-the-art rejection and inspection solutions. Their cutting-edge technology helps pharmaceutical manufacturers meet stringent industry standards while optimizing production lines. With automated rejection systems, manufacturers can not only ensure regulatory compliance but also enhance operational efficiency and brand trust. In an industry where patient safety is paramount, pharmaceutical rejection systems are an indispensable tool, reinforcing the commitment to delivering safe and effective medications to the global market.
The importance of quality assurance in pharmaceutical manufacturing
Quality assurance in pharma manufacturing is a cornerstone of the industry, ensuring that every product meets the highest safety and efficacy standards before reaching patients. Given the critical nature of pharmaceuticals, even minor defects in manufacturing or packaging can lead to severe health risks, regulatory penalties, and loss of consumer trust.
To prevent such issues, pharmaceutical manufacturers must adhere to stringent regulations set by global health authorities, including the FDA (Food and Drug Administration), EMA (European Medicines Agency), and WHO (World Health Organization). These regulatory bodies enforce Good Manufacturing Practices (GMP), which outline strict guidelines on cleanliness, process validation, documentation, and quality control. Compliance with these regulations is not optional—it is mandatory to maintain market approval and avoid costly recalls.
A robust quality assurance (QA) framework plays a crucial role in ensuring compliance and preventing defects. This framework involves:
- Raw Material Inspection: Ensuring all ingredients meet purity and potency standards.
- In-Process Quality Control: Continuous monitoring of production to detect deviations.
- Final Product Testing: Conducting rigorous analytical tests before market release.
- Packaging Integrity Checks: Verifying that packaging prevents contamination and tampering.
Incorporating advanced rejection systems further strengthens quality assurance by automatically removing defective products from the production line. These systems utilize vision inspection, precision weighing, and sensor-based technology to identify inconsistencies, ensuring that only flawless products reach consumers.
With evolving regulations and increasing scrutiny, quality assurance in pharma manufacturing is more important than ever. Investing in advanced QA protocols not only ensures compliance but also enhances operational efficiency, reduces waste, and protects brand reputation. Ultimately, a strong quality assurance system is the foundation of a safe and trustworthy pharmaceutical supply chain.
Common defects in pharmaceutical packaging
In the pharmaceutical industry, rejecting defective pharmaceutical products is a critical step in maintaining safety, compliance, and product efficacy. Packaging defects can happen at various stages of production, and if not identified and removed, they can lead to severe health risks, regulatory violations, and financial losses. Some of the most common packaging defects include:
1. Mislabeling
Incorrect or missing labels can cause serious medication errors, leading to overdoses, adverse drug interactions, or ineffective treatments. A mislabelled product may provide incorrect dosage instructions, lack vital warnings, or misrepresent the active ingredients, putting patient safety at risk.
2. Contamination
Foreign particles, bacteria, or chemical residues in pharmaceutical packaging can render medications unsafe for consumption. Contamination may occur due to inadequate sterilization, compromised manufacturing environments, or packaging material defects. Even minor contamination can lead to infections, allergic reactions, or reduced drug efficacy.
3. Seal integrity issues
Damaged or improperly sealed packaging can expose medications to moisture, air, and contaminants, compromising their stability and shelf life. Weak seals in blister packs, vials, or bottles can lead to product degradation, making the medication ineffective or even harmful to patients.
4. Structural defects
Packaging defects such as cracked bottles, punctured blister packs, or broken caps can impact the protection of the pharmaceutical product. Poor structural integrity increases the risk of leaks, spills, or evaporation of active ingredients, affecting the medication’s potency.
5. Counterfeit or tampered packaging
Tampered seals or counterfeit packaging pose a significant risk in the pharmaceutical supply chain. Patients receiving counterfeit drugs may unknowingly consume ineffective or toxic substances, leading to serious health consequences.

E2M COUTH’s advanced rejection systems for pharmaceutical packaging
E2M COUTH specializes in advanced pharmaceutical inspection and rejection systems, designed to maintain the highest standards in quality control, compliance, and production efficiency. These systems ensure that only flawless products proceed along the manufacturing line, preventing defective packaging from reaching the market.
1. Proportional Rejection Systems
These automated rejection solutions efficiently separate defective pharmaceutical containers based on detected irregularities. Together with high-speed inspection systems, proportional rejection mechanisms ensure that non-conforming products are accurately removed without disrupting the workflow. This is crucial in maintaining regulatory compliance and reducing production waste.
2. Push Rejection Systems
The push rejection technology utilizes a synchronized system composed of a cylinder, electrovalve, and shovel mechanism to divert defective products into a designated rejection area. This method is particularly effective for handling various container types, including bottles, vials, and blister packs, without compromising the integrity of the remaining production batch.
Adaptability across the pharmaceutical production process
E2M COUTH’s inspection and rejection systems are highly versatile and can be seamlessly integrated into multiple stages of pharmaceutical packaging, including:
- Dosing: Ensuring only accurately filled containers proceed.
- Capping & Sealing: Removing containers with faulty closures.
- Filling: Detecting and rejecting improperly filled units.
- Labeling: Eliminating misprinted or mislabeled products.
- Final Packaging: Providing an additional layer of quality assurance before shipment
Optimizing pharmaceutical manufacturing with cutting-edge rejection systems
E2M COUTH’s rejection solutions are engineered to handle the stringent demands of the pharmaceutical industry, offering:
- Precision: High-speed, real-time defect detection and removal.
- Regulatory Compliance: Meets GMP and FDA standards.
- Efficiency: Reduces product loss and enhances operational flow.
- Customization: Adaptable to diverse packaging formats and production speeds.
By implementing E2M COUTH’s advanced rejection technologies, pharmaceutical manufacturers can ensure product integrity, safety, and compliance, reinforcing trust in their brand and regulatory standing.
How E2M COUTH’s inspection and rejection systems prevent packaging defects
Pharmaceutical manufacturers must adhere to strict quality control standards to ensure product safety and regulatory compliance. E2M COUTH’s automated rejection systems play a vital role in identifying and eliminating defective pharmaceutical products, preventing substandard packaging from reaching consumers.
1. Real-time defect detection
E2M COUTH’s automated rejection systems integrate seamlessly with high-speed inspection units to detect anomalies in pharmaceutical packaging. These anomalies can include:
- Incorrect dosing: Overfilled or underfilled containers.
- Sealing and capping errors: Loose caps or improperly sealed blister packs.
- Mislabeling or missing information: Smudged, misprinted, or missing labels.
- Structural defects: Damaged or deformed packaging.
2. Precise defective product elimination
Once an irregularity is detected, E2M COUTH’s rejection systems swiftly remove the defective unit from the production line, ensuring that only flawless products proceed. Key rejection mechanisms include:
- Proportional Rejection Systems: Automatically separates non-compliant units without disrupting the production flow.
- Push Rejection Systems: Uses a synchronized cylinder, electrovalve, and shovel mechanism to divert defective products into a rejection area.
Enhancing Product Quality and Compliance
By integrating these advanced rejection technologies, E2M COUTH helps pharmaceutical manufacturers achieve:
Improved product integrity
Automated rejection ensures that every pharmaceutical package meets high-quality standards, preventing the risk of contamination, mislabeling, or incorrect dosages.
Regulatory compliance
E2M COUTH’s rejection systems help manufacturers comply with Good Manufacturing Practices (GMP), FDA, and European Medicines Agency (EMA) standards, avoiding costly recalls and legal penalties.
Reduction in consumer risk
By eliminating defective products early in the process, manufacturers significantly reduce the risk of distributing non-compliant pharmaceuticals, safeguarding consumer health and brand reputation.
Optimized production efficiency
Automating the rejection process minimizes human intervention, leading to faster production speeds, reduced waste, and lower operational costs.
E2M COUTH’s cutting-edge rejection systems offer pharmaceutical companies unparalleled precision and reliability, ensuring that only safe and compliant products reach the market. By integrating these systems into their production lines, manufacturers can maintain high quality, regulatory adherence, and consumer trust.
Benefits of Implementing E2M COUTH’s Advanced Rejection Technologies
In the pharmaceutical industry, maintaining high product quality and regulatory compliance is crucial. E2M COUTH’s advanced rejection technologies provide a reliable, automated solution to eliminate defective products from the production line, ensuring only flawless packaging reaches consumers.
Increased production efficiency
By integrating automated rejection systems, pharmaceutical manufacturers can:
- Detects and removes defective products in real time, avoiding bottlenecks.
- Minimize manual intervention, reducing human error and improving workflow.
- Maintain high-speed production without compromising quality.
Cost savings from reduced waste
Defective packaging can lead to significant material loss and financial waste. E2M COUTH’s rejection systems help:
- Prevent costly product recalls by ensuring defective items never reach the market.
- Reduce material waste by identifying errors early in the process.
- Optimize resource utilization, cutting down on operational expenses.
Improved brand reputation and consumer trust
Consistently delivering high-quality pharmaceutical products strengthens brand credibility. E2M COUTH’s technology ensures:
- Strict adherence to pharmaceutical regulations (GMP, FDA, EMA).
- Reliable rejection of mislabelled, underfilled, or damaged products.
- A strong reputation for quality, safety, and compliance in the industry.
By adopting E2M COUTH’s advanced rejection systems, pharmaceutical companies can boost efficiency, reduce costs, and enhance brand integrity, ensuring long-term market competitiveness.
Key considerations when selecting rejection systems for pharmaceutical packaging
In this section, we will focus on the critical factors pharmaceutical manufacturers should consider when selecting rejection systems for their packaging lines. These considerations will ensure the chosen system aligns with production goals, enhances quality assurance, and maintains regulatory compliance.
System compatibility with existing equipment
It’s essential to evaluate how well a pharmaceutical rejection system integrates with current packaging lines. Manufacturers should ensure that the rejection system can be seamlessly incorporated into the production process without significant modifications to existing equipment. This compatibility helps minimize downtime and additional training costs for operators.

Detection sensitivity and accuracy
The primary function of rejection systems is to identify defective products quickly and accurately. Manufacturers must assess the detection capabilities of the system, ensuring that it can effectively identify various types of defects such as mislabeling, packaging inconsistencies, and contamination. A highly sensitive detection system will ensure that only compliant products make it to the next stage of the production line.
Speed and Efficiency
In pharmaceutical manufacturing, speed is crucial to maintaining productivity while meeting stringent regulatory standards. Automated rejection systems should operate at high speeds, rejecting defective products without slowing down the overall production process. Ensuring that these systems are both efficient and fast will help minimize product loss and increase production throughput.
Ease of maintenance and support
Another vital consideration is the ease of maintenance and the level of technical support available for the rejection system. Manufacturers should choose systems that are easy to maintain with readily available spare parts. Additionally, the provider’s customer support should be responsive and capable of resolving any operational issues swiftly to avoid disruptions in production.
Regulatory compliance
As pharmaceutical products must adhere to strict regulatory guidelines, it is imperative to choose rejection systems that meet industry standards such as Good Manufacturing Practices (GMP). The selected system must comply with local and international regulations to ensure that all products are produced in accordance with the necessary safety and quality standards.
Cost vs. benefit
Finally, manufacturers must weigh the costs of the rejection system against its potential benefits. While advanced systems may come with higher initial costs, the long-term benefits, such as reduced product recalls, improved quality assurance, and enhanced production efficiency, often outweigh the initial investment. A thorough cost-benefit analysis will help manufacturers make an informed decision that aligns with their budget and operational needs.
E2M COUTH’s pharmaceutical rejection systems are essential in preventing packaging defects, ensuring high-quality standards, and protecting patient safety. Investing in these advanced rejection technologies helps pharmaceutical manufacturers maintain compliance, reduce waste, and uphold their reputation for delivering safe, reliable products.
Need more information? Then contact us and we will help you with the best consulting.